Global Biopharmaceutical Industry Takes Action to Combat Antimicrobial Resistance (AMR)
Published 09-20-16
Submitted by International Federation of Pharmaceutical Manufacturers & Associations
 Delivering on the Industry Declaration
Delivering on the Industry DeclarationIn the side-lines of the United Nations General Assembly High-level Meeting on Antimicrobial Resistance (AMR) in New York, the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) will convene a panel debate to discuss “The Role of Partnerships in Supporting Innovation to Combat Antimicrobial Resistance (AMR)” and report on industry progress in the fight against drug-resistant infections.
Antimicrobials, and specifically antibiotics, play a crucial role in modern medicine. These precious medicines are not only necessary to treat life-threatening infections, but are also vital to underpin most common surgical procedures and many chronic treatments such as chemotherapy and HIV and transplant medicines.
The increase in bacterial resistance to antibiotics has been dramatic, and combating this growth is a top priority for global policy and public health. It is estimated that 10 million people may die every year from bacterial infections by 2050. AMR has been identified as one of the greatest health challenges of our time.
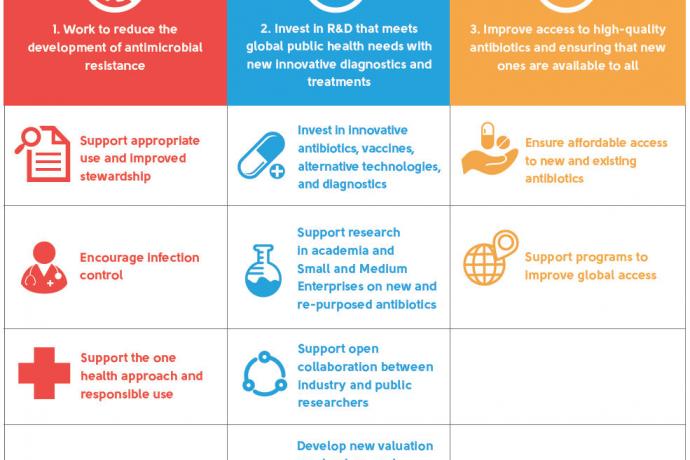
To promote collective action in response to AMR, the pharmaceutical, biotechnology and diagnostics industries launched a joint Declaration on Combating Antimicrobial Resistance. The signatories to the Declaration commit to:
Work to reduce the development of antimicrobial resistance
Invest in R&D that meets global public health needs with new innovative diagnostics and treatments
Improve access to high-quality antibiotics and ensuring that new ones are available to all
“This week's UN High-level Meeting on AMR is a major milestone in galvanizing efforts to combat AMR” – says Mr. Mario Ottiglio, Director, Public Affairs, Communications and Global Health Policy, IFPMA. “The biopharmaceutical industry is more aware than many of the importance of challenges and the Declaration highlights what we can do, but also draws attention to where the support of governments and is needed”.
The IFPMA Health Partnerships Directory lists over 20 collaborative initiatives to combat AMR, demonstrating biopharmaceutical companies’ efforts to deliver on the commitments they made in the Industry Declaration. Partnerships include the Japanese-led Global Health Innovative Technology (GHIT) Fund, with a number of projects developing solutions for AMR, access programs for new antibiotics to combat drug-resistant tuberculosis, and investigational vaccines designed to reduce hospital-acquired infections.
Offering support for a comprehensive and multi-sectoral approach to address the other factors contributing to AMR, as defined by the World Health Organization Global Action Plan, and building off the aforementioned Declaration, 13 leading biopharmaceutical companies today announce a new roadmap that lays out four commitments to reduce AMR by 2020: reducing manufacturing pollution, addressing inappropriate use, improving global access and developing a broad R&D ecosystem.
“The Declaration and the related Roadmap demonstrate the commitment of biopharmaceutical companies to continue to seek engagement with stakeholders to progress important issues, including resolving the economic challenges.” – says, Dr. Isao Teshirogi, Vice President, IFPMA and President and CEO, Shionogi & Co. Ltd.
IFPMA will continue to play a pivotal role in supporting and coordinating Declaration signatories’ activities and stakeholder engagement in this area.
About IFPMA
IFPMA represents research-based biopharmaceutical companies and associations across the globe. The research-based biopharmaceutical industry’s 2 million employees research, develop and provide medicines and vaccines that improve the life of patients worldwide. Based in Geneva, IFPMA has official relations with the United Nations and contributes industry expertise to help the global health community find solutions that improve global health.

International Federation of Pharmaceutical Manufacturers & Associations
International Federation of Pharmaceutical Manufacturers & Associations
The International Federation of Pharmaceutical Manufacturers & Associations (IFPMA) represents research-based pharmaceutical companies and associations across the globe. Based in Geneva, IFPMA has official relations with the United Nations and contributes industry expertise to help the global health community find solutions that improve global health.
Research-based pharmaceutical companies make a unique contribution to global health as innovators of life-saving and life-changing medicines and vaccines, which improved millions of lives around the world. Both IFPMA and its companies are involved in hundreds of partnerships, including the Access Accelerated initiative, a partnership of over 20 biopharmaceutical companies developing innovative and sustainable solutions to improve access to non-communicable disease (NCDs) treatment and care in low- and middle income countries. Access Accelerated also partners with the World Bank and the Union of International Cancer Control to help address the full spectrum of access barriers to NCD medicines. These partnerships involve the discovery of treatments that threaten health security or unduly put a burden on low-income countries, as well as support Universal Health Coverage (UHC) by expanding access to medicines and vaccines and strengthening health systems and in doing so, contribute to the Sustainable Development Goals (SDG 3 and SDG 17).
IFPMA hosts the Secretariat of the AMR Industry Alliance, a coalition of over 100 biotechnology, diagnostic, generics and research-based biopharmaceutical companies and trade associations that was formed to drive and measure industry progress to curb antimicrobial resistance. IFPMA also played a key role in establishing the AMR Action Fund, a ground-breaking initiative involving over 20 biopharmaceutical companies, that aims to bring 2-4 new antibiotics to patients by 2030.
Other initiatives supported by IFPMA include: The African Global Health Leaders Fellowship, the Africa Young Innovators for Health Award, and Fight the Fakes.
More from International Federation of Pharmaceutical Manufacturers & Associations

